
For around 20-30 years, PCR has been extensively utilized in research and medicine to discover genetic information. RT-PCR is a modified form of the method that is utilized when RNA is identified, and it is currently being used to identify SARS-CoV-2, the virus that causes COVID-19. This sort of rapid antigen tests has commonly been employed as a first-line diagnostic for COVID-19 because it detects the viral RNA directly.
RT-PCR assays are relatively rapid, sensitive, and reliable, with findings available in 3-4 hours, however, this time is often extended if samples must first be forwarded to specialized external labs (6-8 hours on average).
Numerous diagnostic and research businesses manufacture RT-PCR products, tests, and equipment, ensuring widespread availability of the technique. Certain RT-PCR assays are produced as an ‘all-in-one’ kit, which minimizes laboratory handling and contamination risk.
How it operates
After collecting a sample, chemicals are employed to deplete it of proteins, lipids, and other components, leaving just RNA. This will be a combination of the individual’s genetic material and any viral RNA that may be present.
The enzymes in the rapid antigen tests kit convert the RNA to DNA, which is amplified to enable virus identification using a PCR machine that cycles the rapid antigen tests temperature in such a way that about 35 billion copies of viral DNA are produced for each viral RNA strand that was initially present.
Typically, fluorescent markers are employed to attach to amplified DNA and generate light that can be read by the machine to provide the rapid antigen tests result. A positive rapid antigen tests is one in which the intensity of the light emitted inside the sample exceeds a predetermined threshold. The number of PCR temperature cycles necessary to attain the fluorescence threshold is recorded, providing an estimate of the viral load in the patient sample.
What are rapid antigen tests designed to detect?
RT-PCR is used to determine whether or not viral RNA is present in patient samples. This is accomplished by encoding and amplifying segments of the virus’s genetic material, often the Spike protein, the N protein, or the Envelope (see picture).
To quantify viral RNA, it is transformed to DNA, duplicated several times using multiple temperature cycles in a PCR machine, and then detected using fluorescent markers. If the quantity of fluorescence increases over a specific threshold, this indicates the presence of the virus. The number of temperature cycles required for the machine to attain this threshold is recorded in order to determine the amount of virus contained in the patient sample. The fewer cycles, the greater the amount of virus present. Typically, these samples are obtained by swabbing the nose or throat with either long or short swabs, although they may also be obtained in other methods. Collecting samples from areas where the virus is shed or proliferating increases the rapid antigen tests‘ accuracy. Click here to read how to use a rapid antigen test kit.
What does the outcome imply?
When conducted on a sample from an infected area of the body during an active illness, an RT-PCR rapid antigen tests is very sensitive and moderately reliable.
A positive PCR result indicates that the individual from whom the sample was collected is presently infected with the virus.
Negative PCR result: – A negative PCR result may indicate that the individual is not presently infected with this virus, the virus is not present at the place from where the sample was collected, the sample was of low quality, or the infection is too early or late to detect the replicating virus. This is why negative test findings need the collection of fresh patient samples a few days later to eliminate the possibility of missing an infected individual.
The RT-PCR rapid antigen tests cannot determine if a person had the virus and subsequently cleared it after the end of the COVID-19 sickness, or whether a person had the disease since it detects an only active virus.
Advantages and disadvantages
- RT-PCR is widely acknowledged by scientists and medical personnel as a reliable and well-documented technology.
- Because RT-PCR is so widely used in research and medicine, the technology for rapid antigen tests for COVID-19 is already in place.
- RT-PCR may identify active illness infections, enabling medical personnel to decide who is infected and who is not.
Disadvantages:
- Because RT-PCR is based on collecting and identifying viruses, it may overlook individuals who have cleared the virus and recovered from sickness. learn more ways of recovering form sickness at https://achs.edu/blog/2018/02/27/how-to-recover-from-illness-faster/
- Because viral distribution in the respiratory tract differs across individuals, even if a person is infected, the virus may be detected only in sputum or a nasopharyngeal swab, but not always in both sites concurrently.
- Real-time polymerase chain reaction (RT-PCR) for COVID-19 can only determine whether a person is presently infected with this specific coronavirus. It is unable to offer information on further illnesses or symptoms.
Antigen examinations
Rapid and precise rapid antigen tests is critical for the containment of a highly infectious virus such as SARS-CoV-2. While PCR tests are reliable, they might take a long time to complete. While rapid antigen tests, the second primary form of coronavirus test, are much quicker, they are also significantly less accurate.
Antigens are substances that elicit an immunological response in the body — they initiate the production of antibodies. These assays look for antigens from the SARS-CoV-2 virus using laboratory-made antibodies.
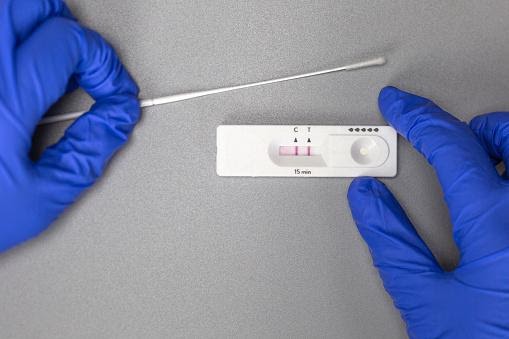
To conduct an antigen test, a sample is first treated with a solution comprising salt and soap, which dissolves cells and other particles. Then you add this liquid to a test strip that has been coated in a narrow line with antibodies specific for SARS-CoV-2.
Antibodies on the test strip, like those in your body, will bind to any antigen in the sample. If the antibodies recognize coronavirus antigens, a colored line indicates the presence of SARS-CoV-2 on the rapid antigen tests strip.
Antigen testing provides a variety of advantages. To begin, they are so simple to do and interpret that they may be performed and interpreted by someone with no particular training – even at home. Additionally, they provide effects swiftly, generally within 15 minutes. Another advantage is that these rapid antigen tests are quite affordable, costing between $10 and $15 for each test.